The Science Behind MolecuLight®: A Wealth of Clinical Data
Featured MolecuLight Publications
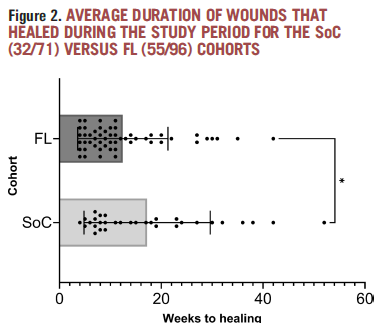
Improving Wound Healing and Infection Control in Long-term Care with Bacterial Fluorescence Imaging
Kelso, MR et al. Adv Skin & Wound Care 2024
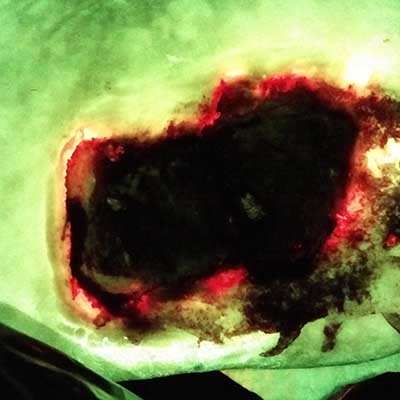
The Use of Point-of-Care Bacterial Autofluorescence Imaging in the Management of Diabetic Foot Ulcers: A Pilot Randomized Controlled Trial
Rahma, S. et al. Diabetes Care 2022
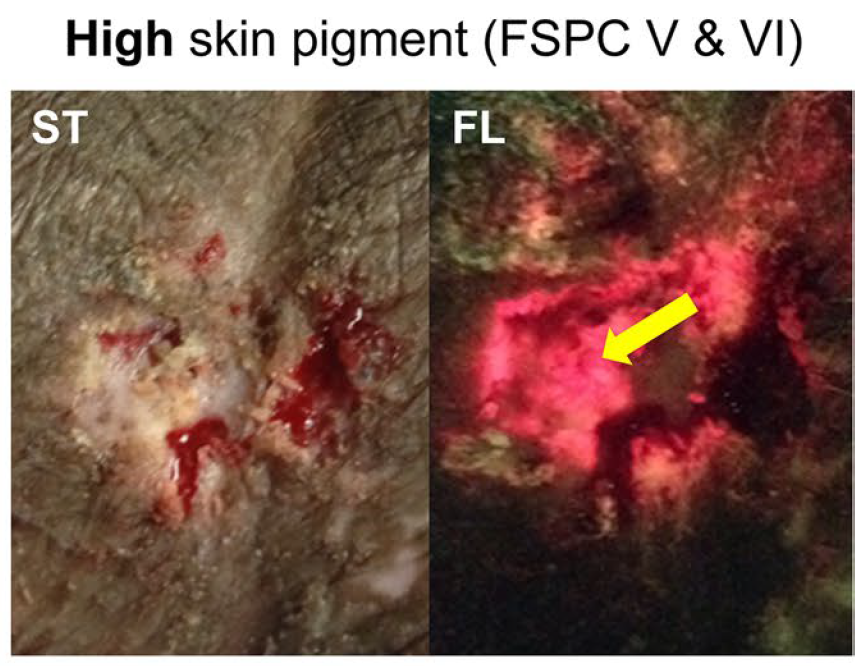
Skin Pigmentation Impacts the Clinical Diagnosis of Wound Infection: Imaging of Bacterial Burden to Overcome Diagnostic Limitations
Johnson J. et al. JREHD 2023
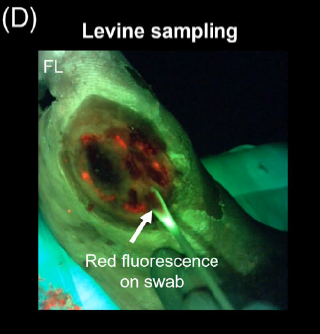
Lights, fluorescence, action—Influencing wound treatment plans including debridement of bacteria and biofilms
Jacob, A et al. Int Wound J 2023
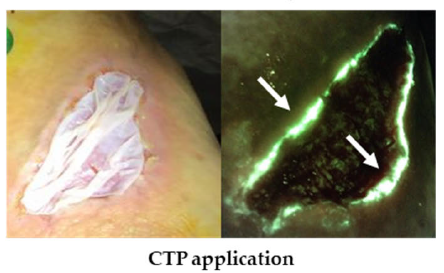
Bacterial fluorescence imaging as a predictor of skin graft integration in burn wounds
Hanson-Viana, E. et al. Burns 2024
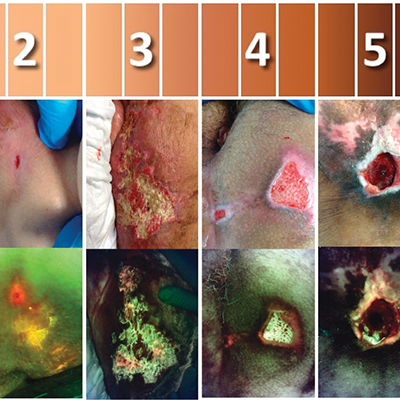
Guidelines for Point-of-Care Fluorescence Imaging for Detection of Wound Bacterial Burden Based on Delphi Consensus
Oropallo A. et al. Diagnostics, 2021
Bacterial Fluorescence Imaging to Address Racial Inequities in Wound Infection Assessment
Andersen, CA et al. Adv Skin & Wound Care 2024
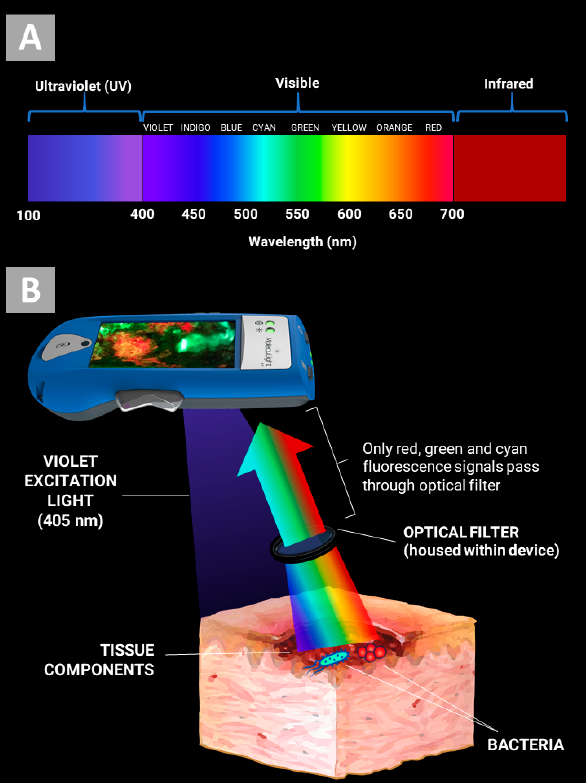
Understanding Real-Time Fluorescence Signals from Bacteria and Wound Tissues Observed with the MolecuLight i:X
Rennie, MY et al. Diagnostics 2023
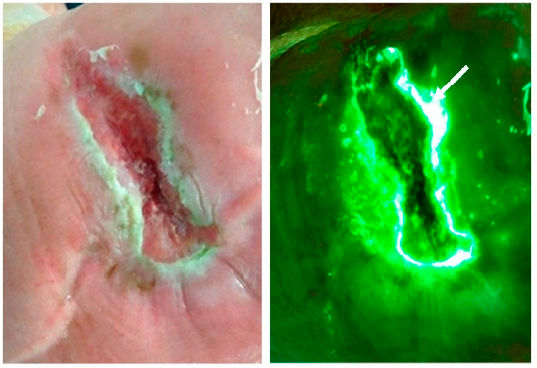
Routine fluorescence imaging to detect wound bacteria reduces antibiotic use and antimicrobial dressing expenditure while improving healing rates: Retrospective analysis of 229 foot ulcers
Price, N. et al. Diagnostics 2020
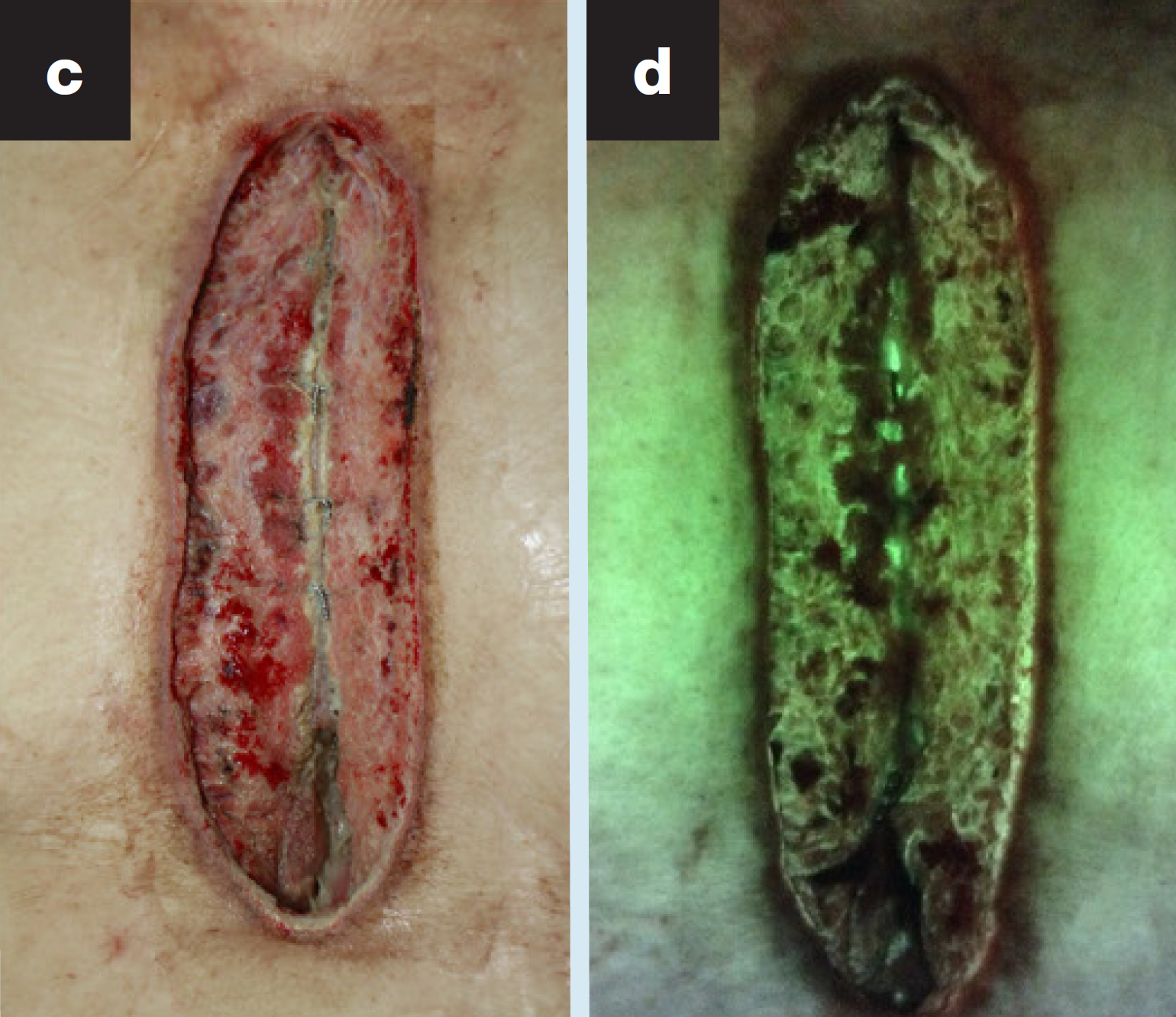
Effects of adjuvant hyperbaric oxygen therapy and real-time fluorescent imaging on deep sternal wound infection: a retrospective study
Li T-H. et al. J Wound Care 2025
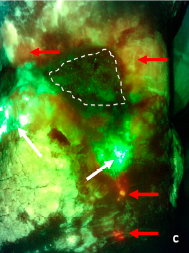
Integrating Point-of-Care Bacterial Fluorescence Imaging-Guided Care with Continued Wound Measurement for Enhanced Wound Area Reduction Monitoring
Derwin, R. et al. Diagnostics 2024
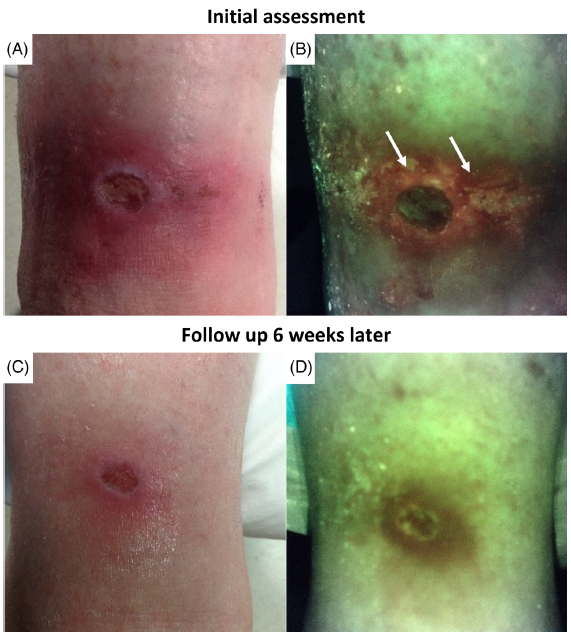
Diagnosis and treatment of the invasive extension of bacteria (cellulitis) from chronic wounds utilizing point-of-care fluorescence imaging
Andersen, A. et al. Int Wound J 2021
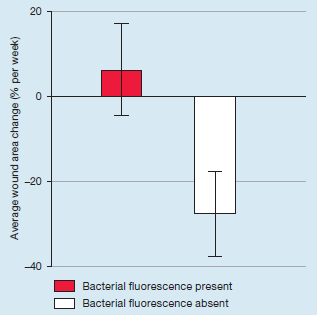
Use of a bacterial fluorescence imaging system to target wound debridement and accelerate healing: a pilot study
Cole, W and Coe S. J Wound Care 2020
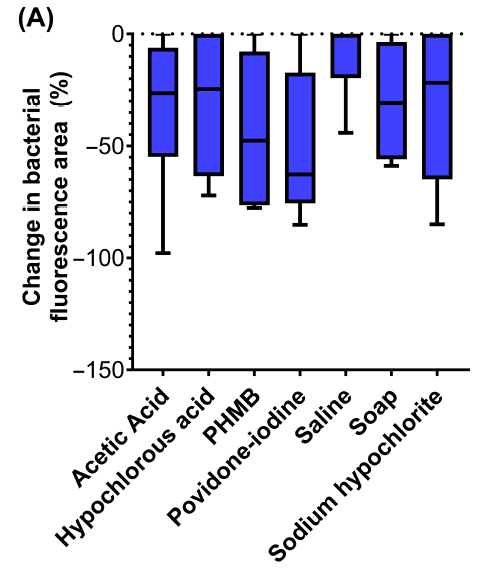
An objective comparative study of non-surgical cleansing techniques and cleanser types in bacterial burden management
Oropallo, A. et al. Int Wound J 2024
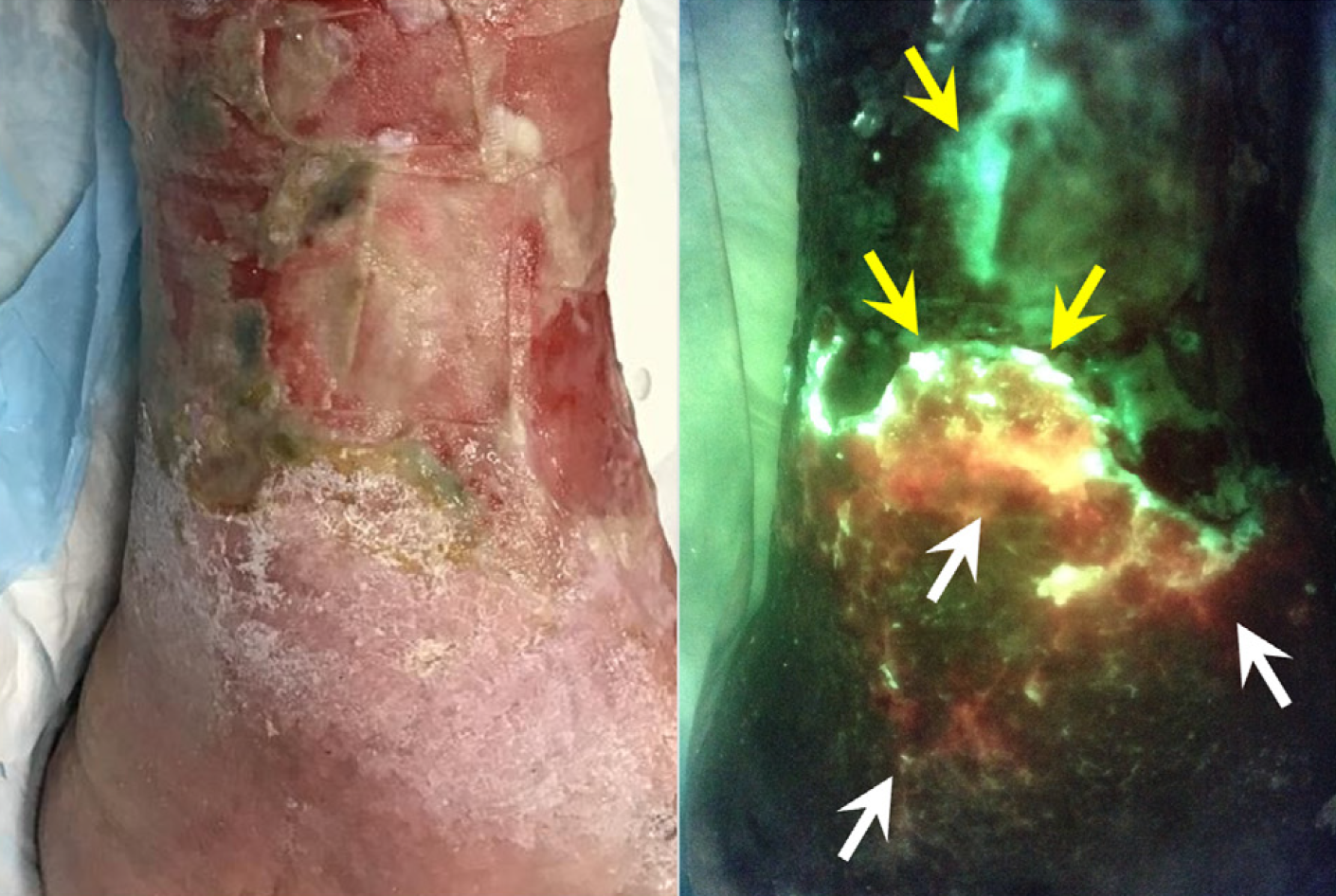
Unveiling the relationship between pain and bacterial load in venous ulcers with implications in targeted treatment
Oropallo A. et al. JVS-VL 2025
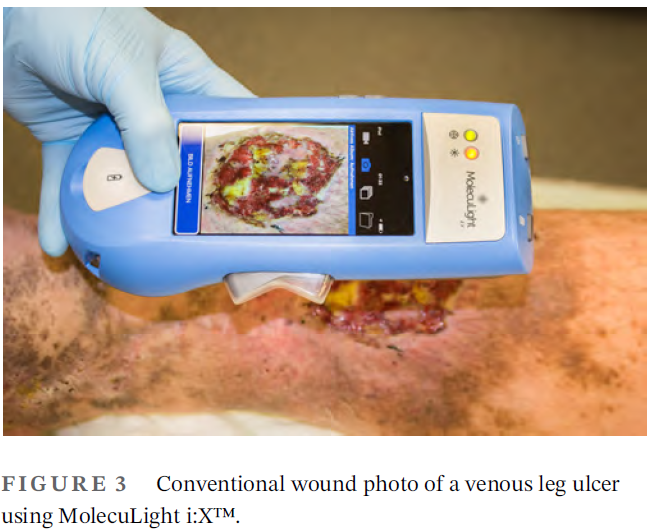
How effective is simple mechanical wound debridement in reducing bacterial colonisation? Results of a prospective clinical study
Moelleken, M et al. Int Wound J 2024
A novel debridement device for the treatment of hard-to-heal wounds: a prospective trial
Al-Jalodi, O et al. J Wound Care 2021
Prospective clinical study on the efficacy of bacterial removal with mechanical debridement in and around chronic leg ulcers assessed with fluorescence imaging
Moelleken, M et al. Int Wound J 2020
Imaging of bacteria in burn wounds treated with split thickness grafts in MEEK/MESH technique: a pilot study with first experiences in clinical wound evaluation with autofluorescence
Alawi, SA et al. HMPC 2019 (Article in German)
Utility of MolecuLight i:X for managing bacterial burden in pediatric burns
Farhan, N et al. J Burn Care Research 2020

Assessing Pediatric Burn Wound Infection Using a Point-of-Care Fluorescence Imaging Device
Turner, E et al. J Burn Care Res 2024
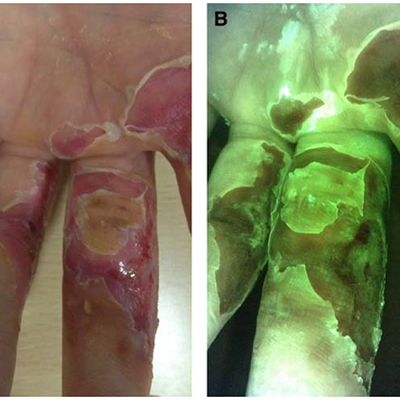
The use of the MolecuLight i:X in managing burns: a pilot study
Blumenthal, E et al. J Burn Care Research 2018
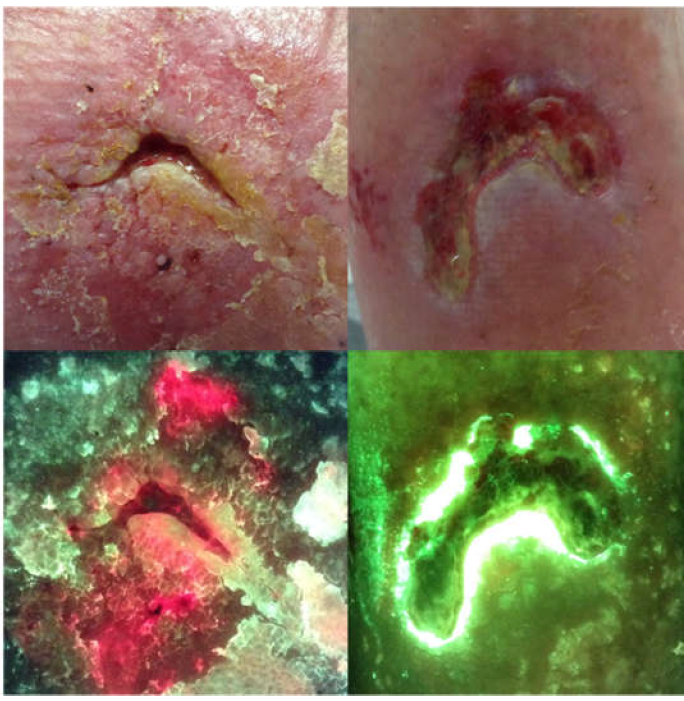
Diagnosing burn wounds infection: The practice gap & advances with MolecuLight bacterial imaging
Farhan, N. et al. Diagnostics 2021
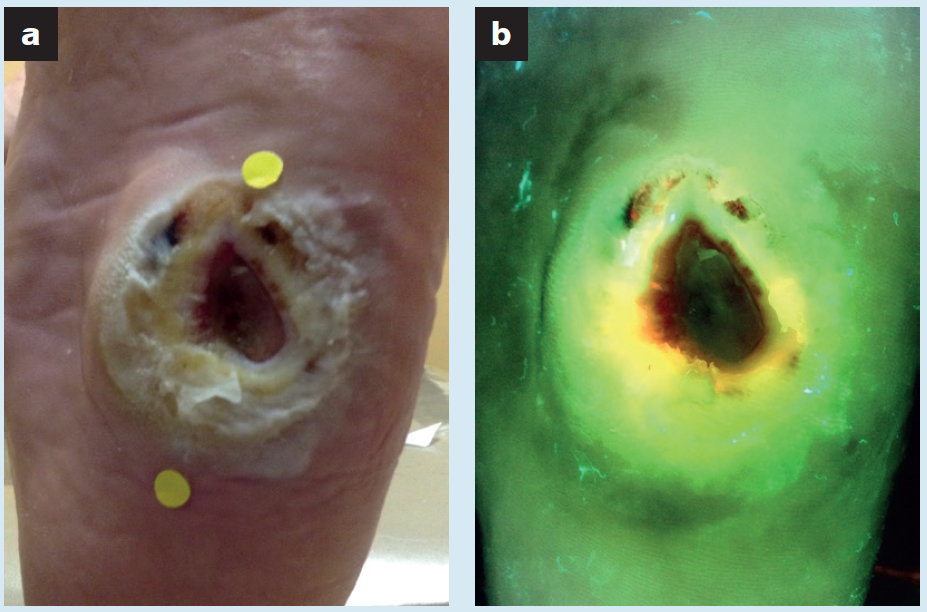
Efficacy and safety of a porcine peritoneum-derived matrix in diabetic foot ulcer treatment: a pilot study
Ai-Jalodi, O et al. J Wound Care 2021
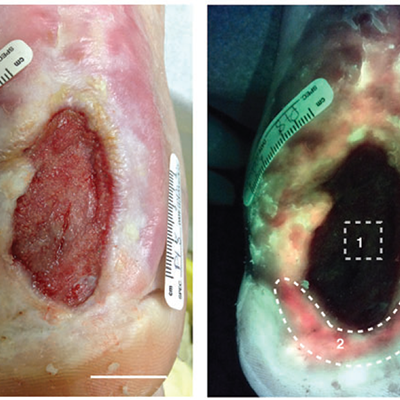
Improved detection of clinically relevant wound bacteria using autofluorescence image-guided sampling in diabetic foot ulcers
Ottolino-Perry, K et al. Int Wound J 2017
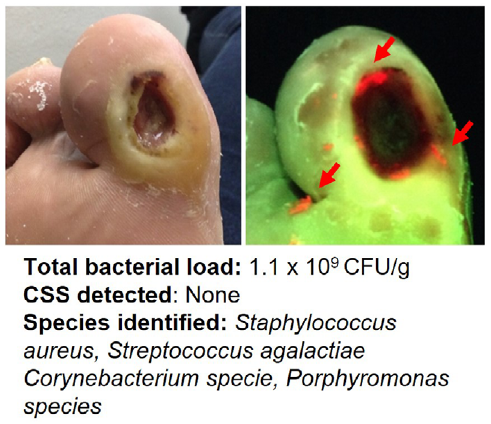
Point-of-care fluorescence imaging reveals extent of bacterial load in diabetic foot ulcers
Armstrong, DG et al. Int Wound J 2023
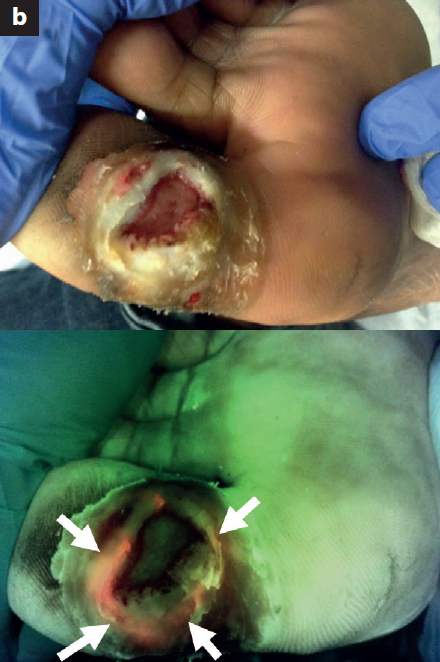
Use of a bacterial fluorescence imaging device: wound measurement, bacterial detection, and targeted debridement
Raizman, R et al. J Wound Care 2019
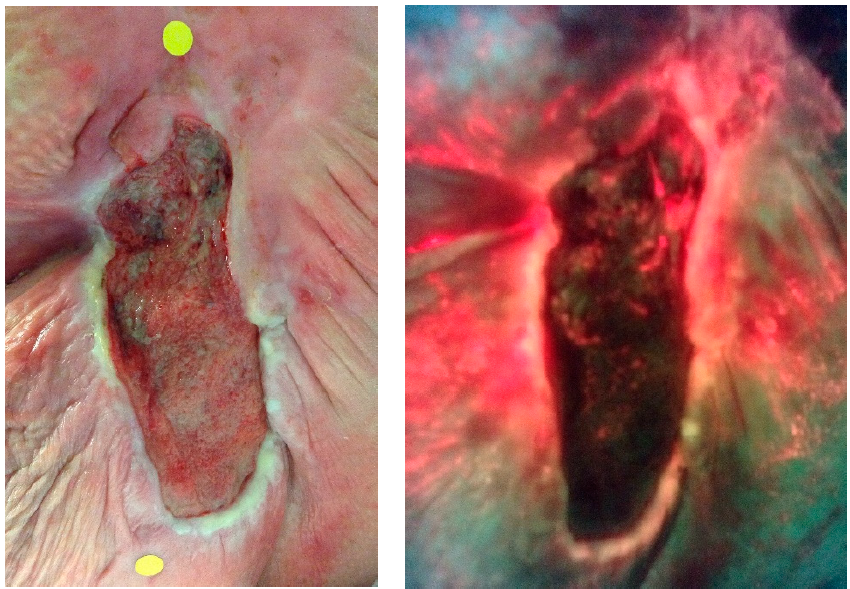
A Diagnostic-Driven Prospective Clinical Study Evaluating the Combination of an Antibiofilm Agent and Negative Pressure Wound Therapy
Serena, TE et al. Diagnostics 2024
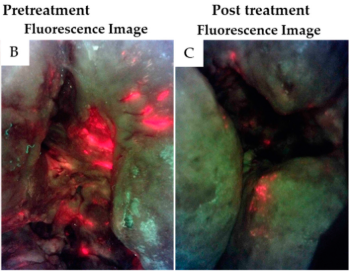
Bacterial Autofluorescence Digital Imaging Guides Treatment in Stage 4 Pelvic Pressure Injuries: A Preliminary Case Series
Stiehl, J.B. Diagnostics 2021
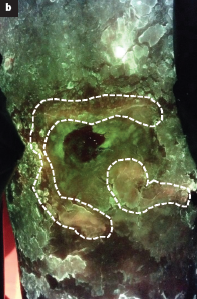
Real-time bacterial fluorescence imaging accurately identifies wounds with moderate-to-heavy bacterial burden
Serena, TE et al. J Wound Care 2019
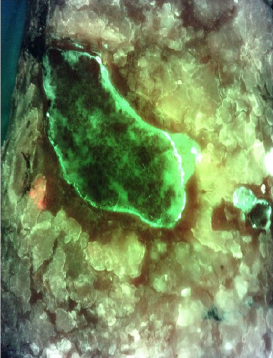
Fluorescence-Based Evaluation of Bacterial Load in Perilesional Skin: A Comparison Between Short Stretch Bandage and Zinc Oxide Bandage
Michelucci, A et al. Int J Low Extrem W 2023
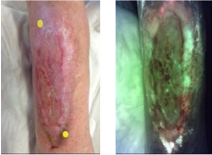
The Role of Autoflorescence Imaging Device in the Evaluation of Bacteria Burden Control
Janowska, A et al. Int J Low Extrem W 2022
Bacterial autofluorescence in infected perineal wounds: A prospective cohort study
Okeahialam NA et al. DMID 2022
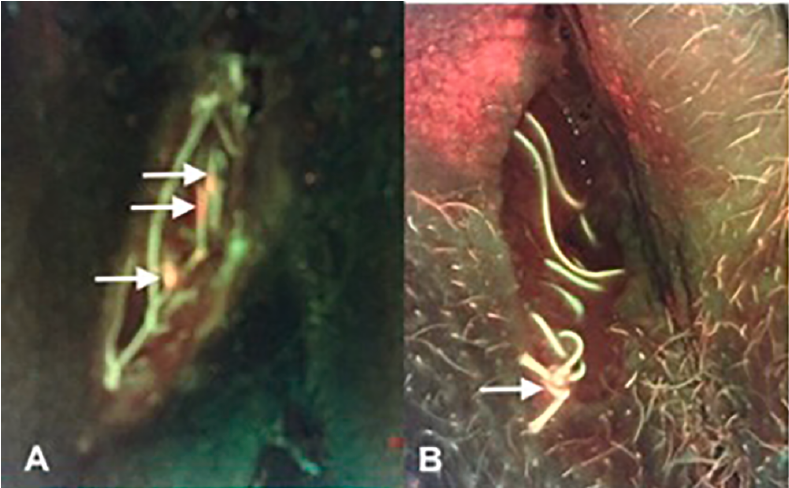
The clinical progression and wound healing rate of dehisced perineal tears healing by secondary intention: A prospective observational study
Okeahialam NA et al. Eur J OGRB 2022
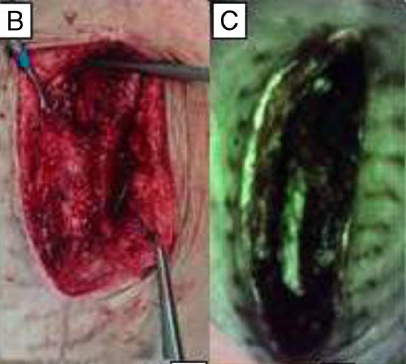
Multidisciplinary Strategies With Real-Time Fluorescence Images and Negative Pressure Wound Therapy to Manage Organ/Space Surgical Site Infection in Transplanted Kidneys
Chen, CH et al. Annals Plastic Surgery 2023
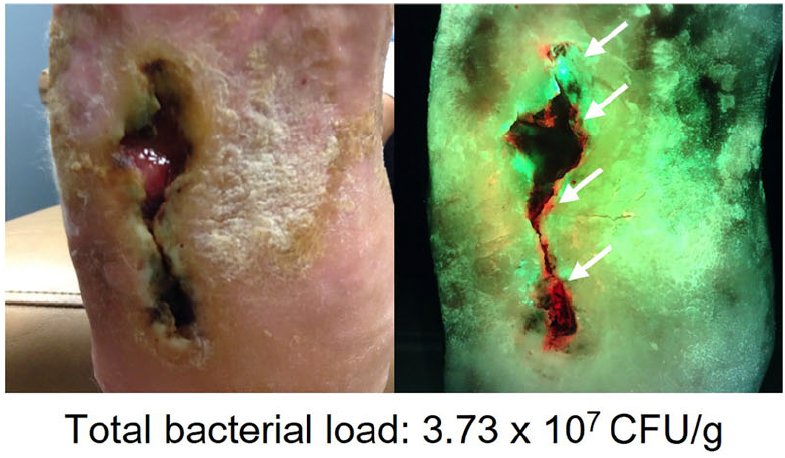
Uncovering the high prevalence of bacterial burden in surgical site wounds with point-of-care fluorescence imaging
Sandy-Hodgetts, K et al. Int Wound Journal 2021

Audit of Antimicrobial Prescribing Trends in 1447 Outpatient Wound Assessments: Baseline Rates and Impact of Bacterial Fluorescence Imaging
Trafelet, N. et al. Diagnostics 2024
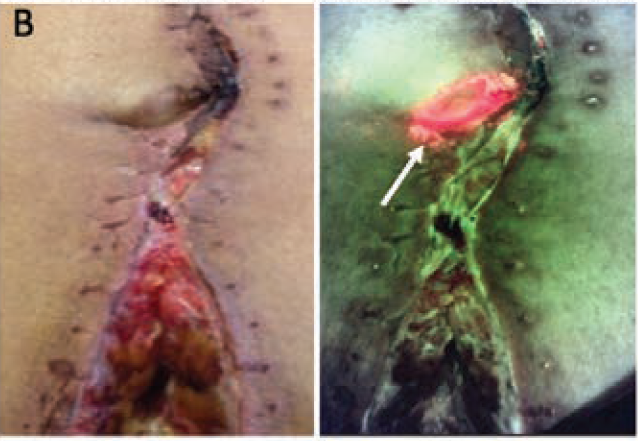
Use of a Fluorescence Imaging Device to Detect Elevated Bacterial Loads, Enhance Antimicrobial Stewardship, and Increase Communication Across Inpatient Complex Wound Care Teams
DasGupta, T et al. Wounds 2022
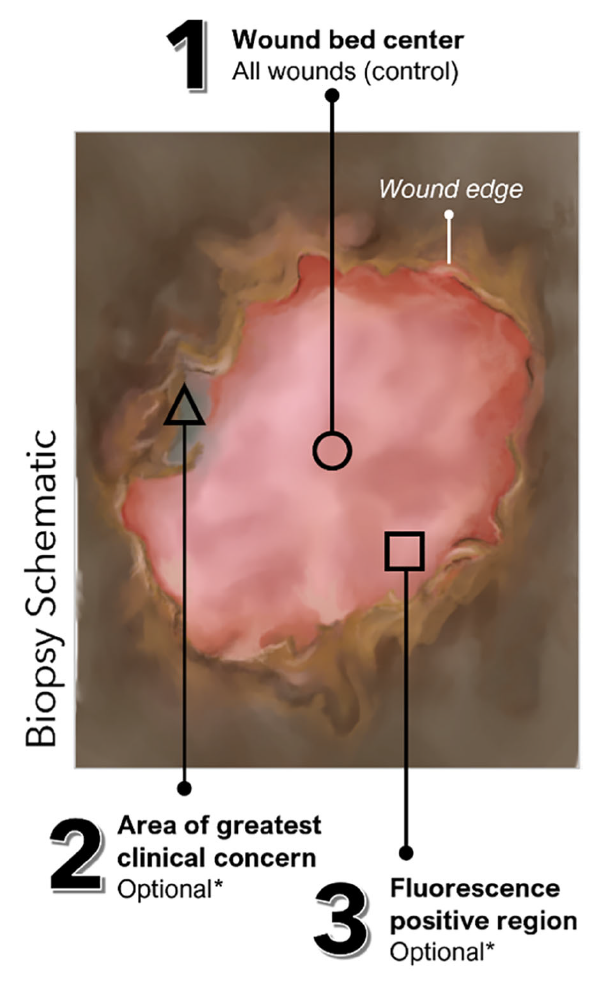
Use of fluorescence imaging (FL) to optimize location of tissue sampling in hard-to-heal wounds
Serena, TE et al. Front Cell Infect Microbiol 2022
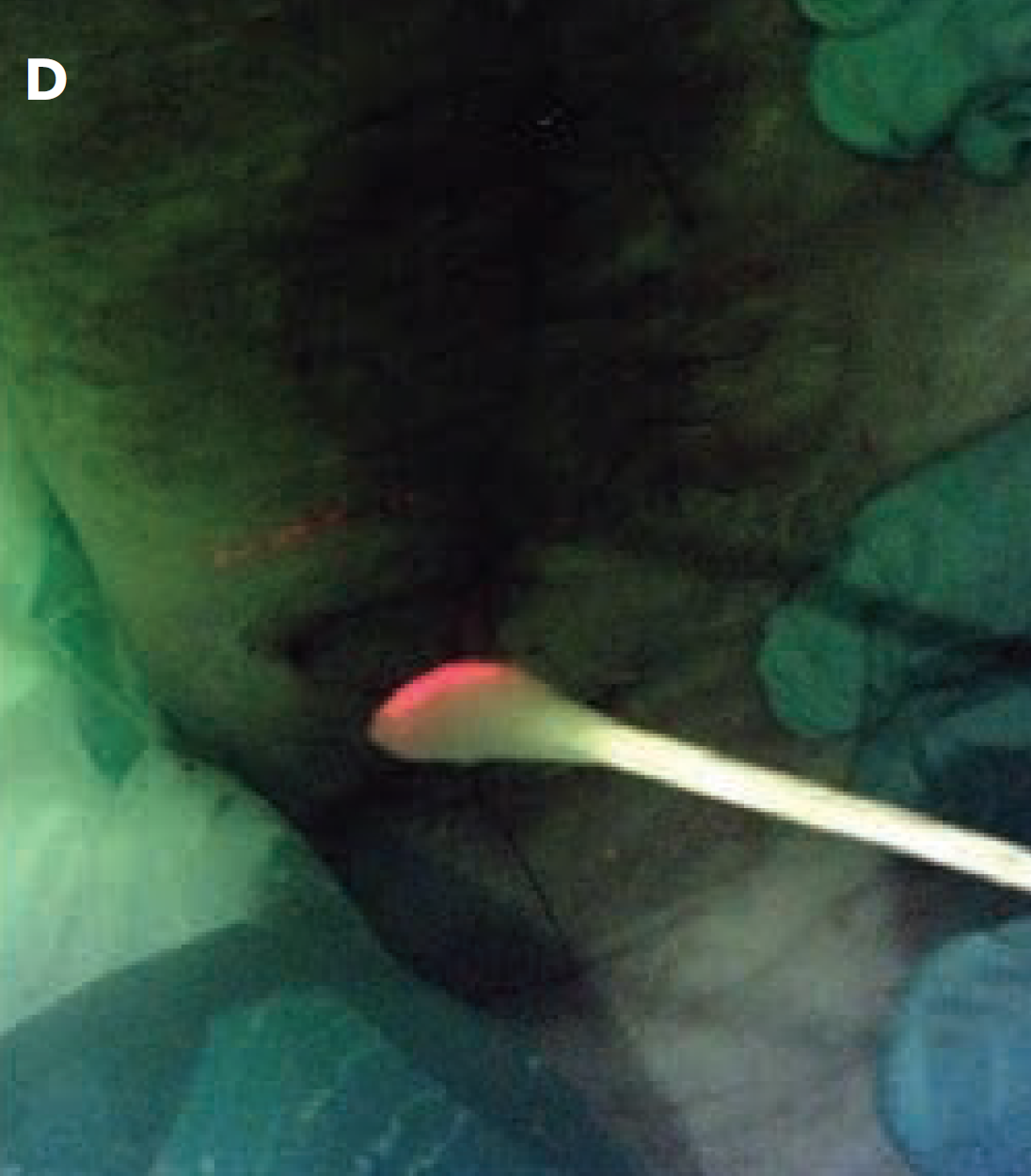
The “C-Swab” test: a technique for identifying bacteria in sinus tracts or tunneled wounds utilizing a cotton swab and bacterial fluorescence imaging
Andersen C. and McLeod K. Wounds 2025

Antibiotic Misuse in Wound Care: Can Bacterial Localization through Fluorescence Imaging Help?
Caputo, WJ et al. Diagnostics (Special Issue) 2022
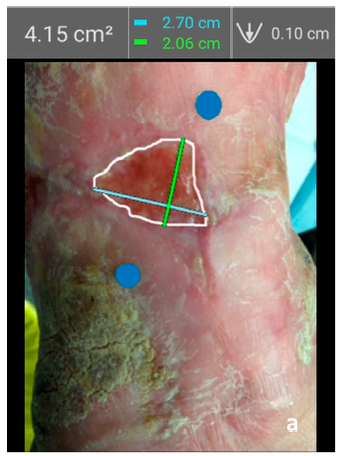
Integrating Point-of-Care Bacterial Fluorescence Imaging-Guided Care with Continued Wound Measurement for Enhanced Wound Area Reduction Monitoring
Derwin, R. et al. Diagnostics 2024

Validation of a Stickerless, Digital Automatic-Depth Measurement Feature within a Multi-Modal Wound Imaging Device
Dunham, D. et al. Presented at SAWC Fall 2024
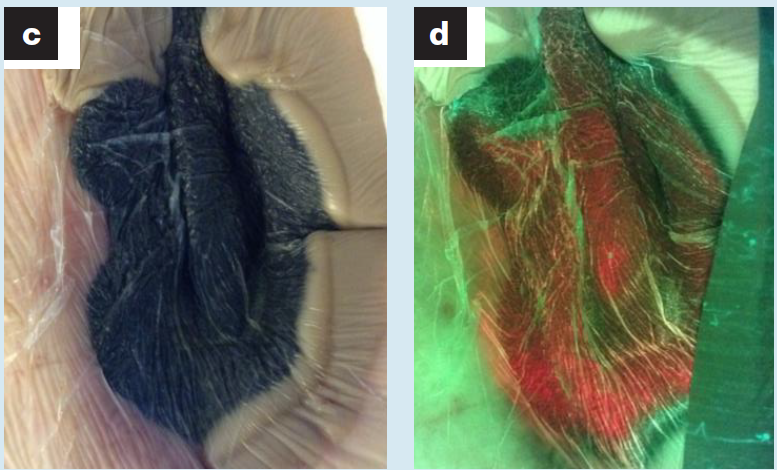
Fluorescence imaging guided dressing change frequency during negative pressure wound therapy: a case series
Raizman, R J. Wound Care 2019
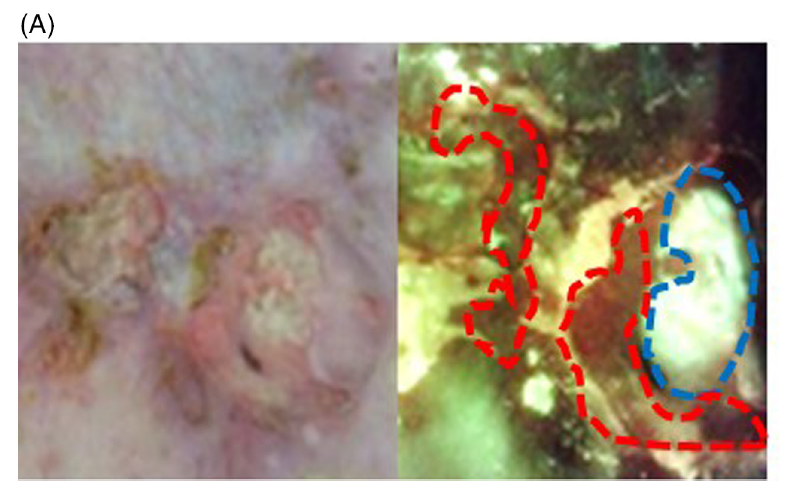
Instantly evaluating bacterial infections on skin ulcers in an Asian population using a fluorescence-emitting device
Kurokami, Y et al. J Cutan Imminol Allergy 2023
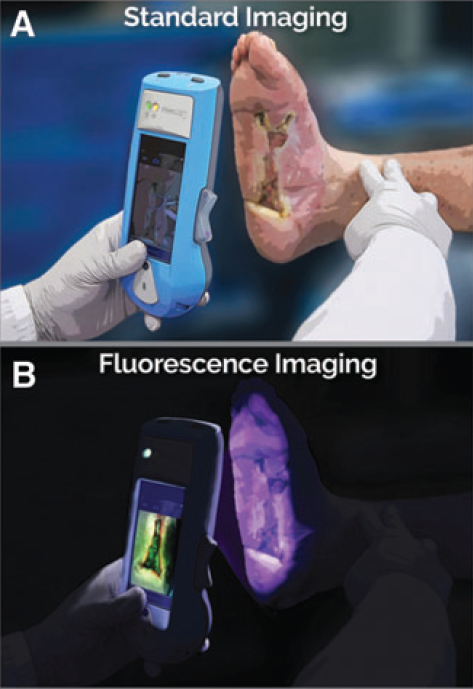
Diagnostic accuracy of point-of-care fluorescence imaging for the detection of bacterial burden in wounds: Results from the 350-patient FLAAG trial
Le, L et al. Adv Wound Care 2021

Diagnostic accuracy of point-of-care fluorescence imaging for the detection of bacterial burden in wounds: Results from the 350-patient FLAAG trial
Le, L et al. Adv Wound Care 2021
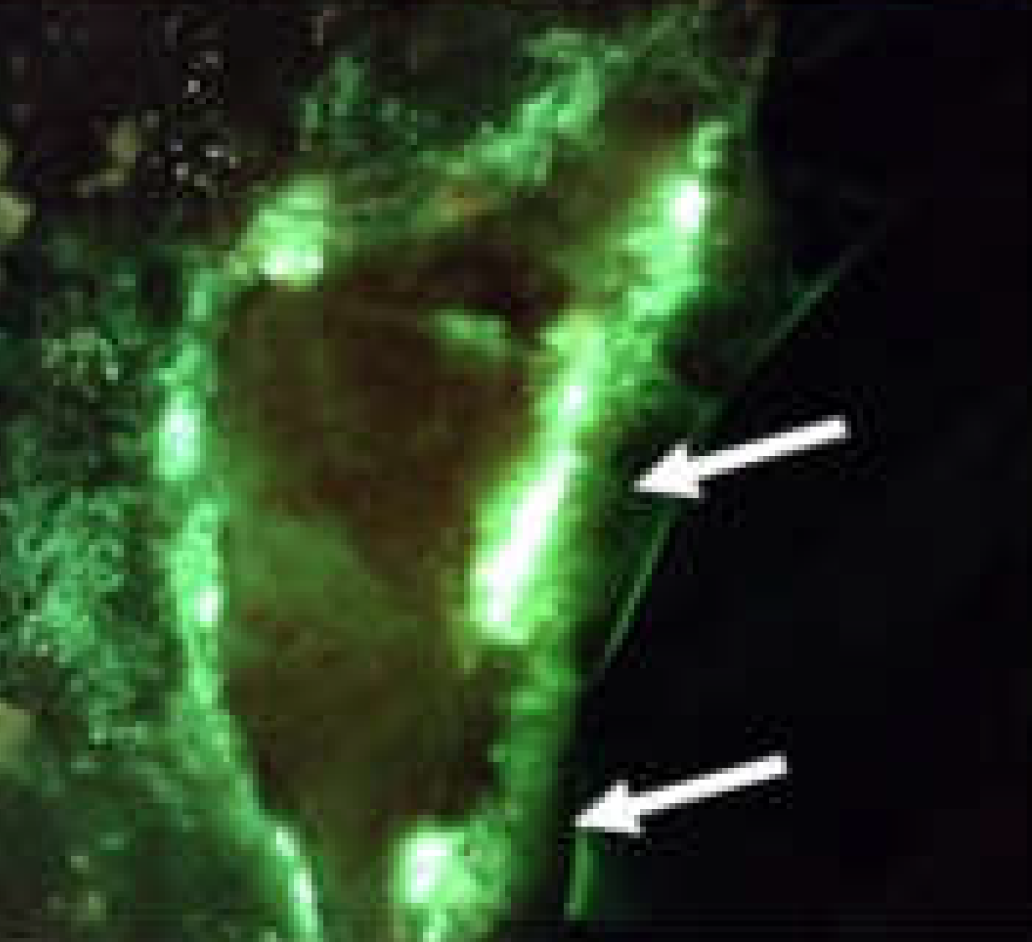
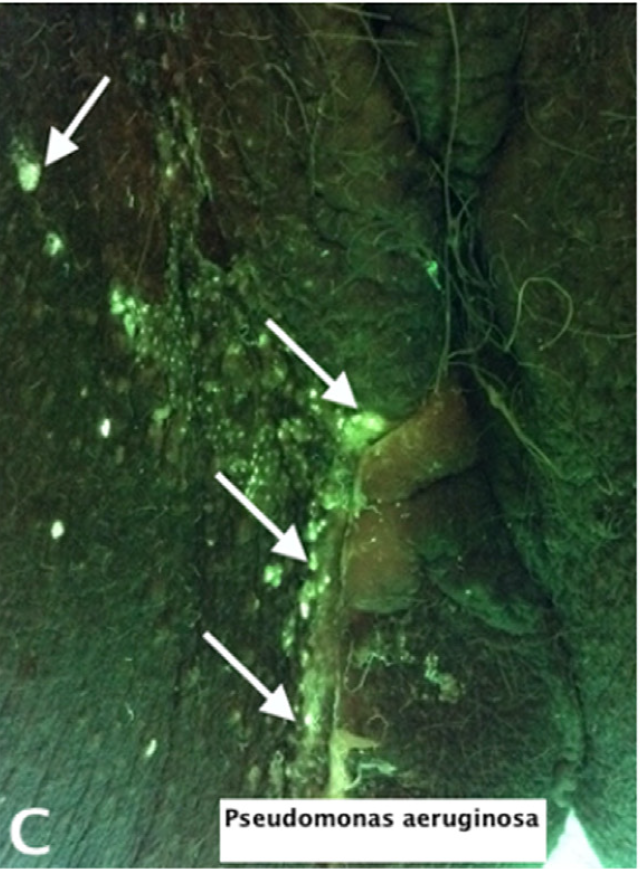
Rapid diagnosis of Pseudomonas aeruginosa in wounds with point-of-care fluorescence imaging
Raizman, R et al. Diagnostics 2021
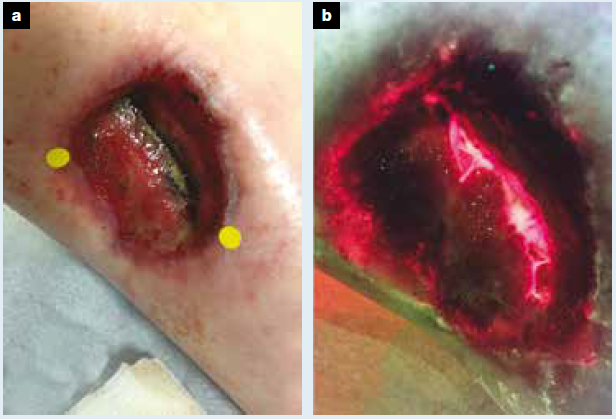
Efficacy of a bacterial fluorescence imaging device in an outpatient wound care clinic: a pilot study
Hurley, CM et al. J Wound Care 2019
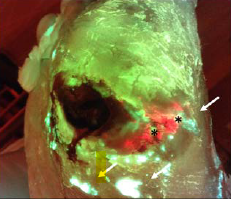
A prospective multi-site observational study incorporating bacterial fluorescence information into the UPPER/LOWER wound infection checklists
Hill, R and Woo, K. Wounds 2020
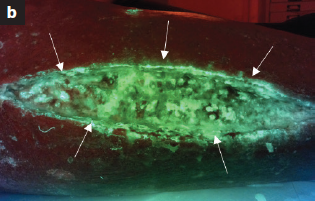
Efficacy of an imaging device at identifying the presence of bacteria in wounds at a plastic surgery outpatients clinic
Blackshaw, EL et al. J Wound Care 2018










