Educational Videos
on Reimbursement & Documentation
View a variety of short instructional videos answering key questions about coding, documentation and reimbursement for the MolecuLight procedure for “fluorescence wound imaging for bacterial presence, location, and load”1,2.
A selection of webcasts describing the use and clinical utility of the MolecuLight procedure are also available for viewing.
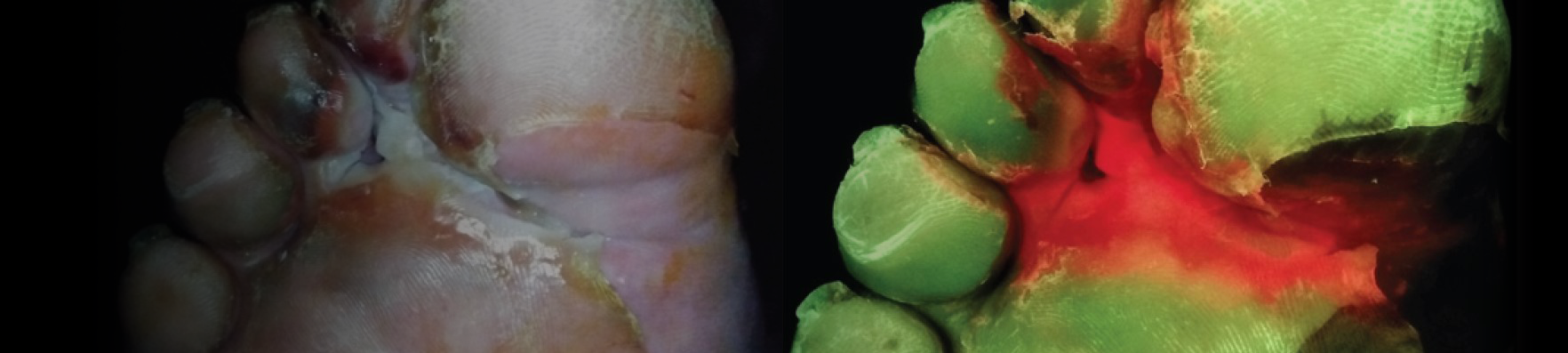
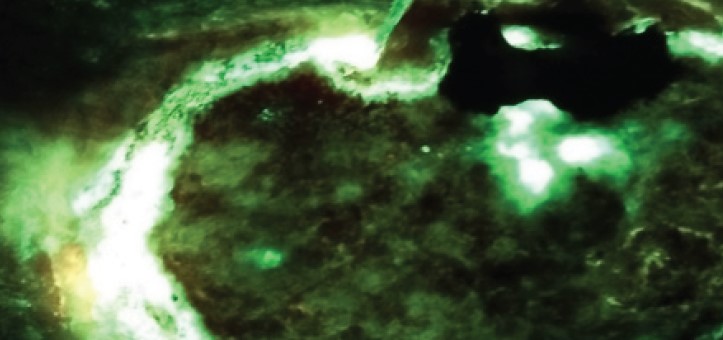
Payer policies will vary and should be verified prior to treatment for limitations on diagnosis, coding, or site of service requirements. The coding options listed here are commonly used codes and are not intended to be an all-inclusive list. Use of the codes provided herein allow payers to track new codes and their associated costs. The MolecuLight i:X detects wounds with elevated loads of bacteria when exposed to an excitation light. Fluorescence is only accurate under clinical darkness achieved using the DarkDrape or turning off the room lights. The MolecuLight i:X does not diagnose or treat skin wounds. The MolecuLight i:X is for prescription use only. This reimbursement guide pertains to the use of MolecuLight i:X in wound care as an imaging device.
DISCLAIMER: This reimbursement guide provides baseline reimbursement information as a convenience to MolecuLight i:X users. It is not a substitute for qualified reimbursement advice as to your specific situation. Coding, coverage and reimbursement decisions are subject to change without notice. Providers should always check with the appropriate payer before submitting claims. The use of appropriate codes, charges for service, and use of modifiers for services rendered is the responsibility of the healthcare provider. Providers are responsible for verifying coverage with payors, including the applicability of any non-coverage policies that may exist, and documentation required as justification/evidence for billed services. MolecuLight Inc. is not responsible for the timeliness, accuracy, completeness, or applicability of the information contained within this document. Since reimbursement laws, regulations, and payor policies change frequently, providers should consult their payers, coding specialists and/or legal counsel regarding coverage, coding and payment issues.
† CPT is a registered trademark of the American Medical Association
References
1 Release of 2 New Category III Codes for Real-time Fluorescence Wound Imaging to the AMA website (Updated July 1, 2020) www.ama-assn.org/system/files/2020-07/cpt-category3-codes-long-descriptors.pdf
2 AMA 2020 CPT Manual all rights reserved.
* The MolecuLight i:X and DX devices have FDA clearance for use for the detection of elevated bacterial burden in wounds when used in combination with clinical signs and symptoms